However heavier pnictogens are more likely to form the +3 oxidation state than lighter ones due to the s-shell electrons becoming more stabilized. When oxidized or ionized, pnictogens typically take an oxidation state of +3 (by losing all three p-shell electrons in the valence shell) or +5 (by losing all three p-shell and both s-shell electrons in the valence shell). The light pnictogens (nitrogen, phosphorus, and arsenic) tend to form −3 charges when reduced, completing their octet. When a pnictogen forms only three single bonds, effects of the lone pair typically result in trigonal pyramidal molecular geometry. Because 5 > 3, it leaves unused two electrons in a lone pair unless there is a positive charge around (like in NH + 4). For example, N 2, the diatomic form of nitrogen, is used as an inert gas in situations where using argon or another noble gas would be too expensive.įormation of multiple bonds is facilitated by their five valence electrons whereas the octet rule permits a pnictogen for accepting three electrons on covalent bonding. Paradoxically, this same strong bonding causes nitrogen's and bismuth's reduced toxicity (when in molecules), because these strong bonds with other atoms are difficult to split, creating very unreactive molecules.

ANTIMONY VALENCE ELECTRONS FREE
When these substances react with various chemicals of the body, they create strong free radicals that are not easily processed by the liver, where they accumulate. This property of these elements leads to their potential toxicity, most evident in phosphorus, arsenic, and antimony. These elements are also noted for their stability in compounds due to their tendency to form covalent double bonds and triple bonds. These compounds are between ionic and covalent compounds and thus have unusual bonding properties. These are in the form of RE aM bPn c, where M is a carbon group or boron group element and Pn is any pnictogen except nitrogen. Other pnictides include the ternary rare-earth main-group variety of pnictides. Pnictide compounds tend to have exotic properties such as being diamagnetic and paramagnetic at room temperature, being transparent, or generating electricity when heated. The most important elements of this group are nitrogen (N), which in its diatomic form is the principal component of air, and phosphorus (P), which, like nitrogen, is essential to all known forms of life.īinary compounds of the group can be referred to collectively as pnictides.

The Russell–Saunders term symbol of the ground state in all elements in the group is 4S 3⁄ 2. They are therefore 3 electrons short of filling their outermost electron shell in their non- ionized state. This group has the defining characteristic that all the component elements have 5 electrons in their outermost shell, that is 2 electrons in the s subshell and 3 unpaired electrons in the p subshell. The "five" ("V") in the historical names comes from the " pentavalency" of nitrogen, reflected by the stoichiometry of compounds such as N 2O 5. In semiconductor physics, it is still usually called Group V. (Pronounced "group five A" and "group five B" "V" is the Roman numeral 5). Deming and the Sargent-Welch Scientific Company, while in Europe it was called Group V B and IUPAC recommended that in 1970.
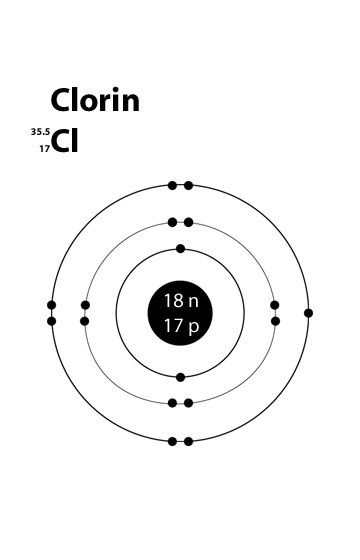
Before that, in America it was called Group V A, owing to a text by H.
Even though unconfirmed, the synthetic element moscovium (Mc) is predicted to be a pnictogen as well. It consists of the elements nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb) and bismuth (Bi). Group 15 is also known as the nitrogen group or nitrogen family. A pnictogen ( / ˈ p n ɪ k t ə dʒ ə n/ or / ˈ n ɪ k t ə dʒ ə n/ from Ancient Greek: πνῑ́γω "to choke" and -gen, "generator") is any of the chemical elements in group 15 of the periodic table.
